Limited Clinical Benefit and Increased Risks: New Review Casts Doubt on Latest Alzheimer’s Drugs
For years, the medical community has held a high level of hope that a new class of antibody treatments could fundamentally change the trajectory of Alzheimer’s disease. However, a comprehensive new analysis has delivered a sobering reality check. A review by the independent scientific network Cochrane suggests that these latest medications offer little to no clinically meaningful benefit for patients, while potentially introducing serious risks.
As Editor-in-Chief of Archysport, I have spent over 15 years reporting on the peak of human performance and the fragility of the human body. Whether covering the Olympic Games or the NFL Super Bowl, the theme is always the same: the data must inform the truth. In this case, the data from a massive review of 17 clinical studies involving more than 20,000 participants indicates that the gap between statistical success and actual patient improvement is wider than previously admitted.
Mehr als 20.000 Patienten, 17 Studien, eine durchwachsene Bilanz: Die neuen Alzheimer-Medikamente zeigen wenig klinisch relevante Wirkung – und könnten teils sogar schaden.
The Gap Between Statistics and Reality
The focus of the controversy centers on monoclonal antibodies, including drugs like Donanemab and Lecanemab. These treatments are designed to target and reduce beta-amyloid, a protein that forms plaques in the brain and is long considered a primary driver of Alzheimer’s.
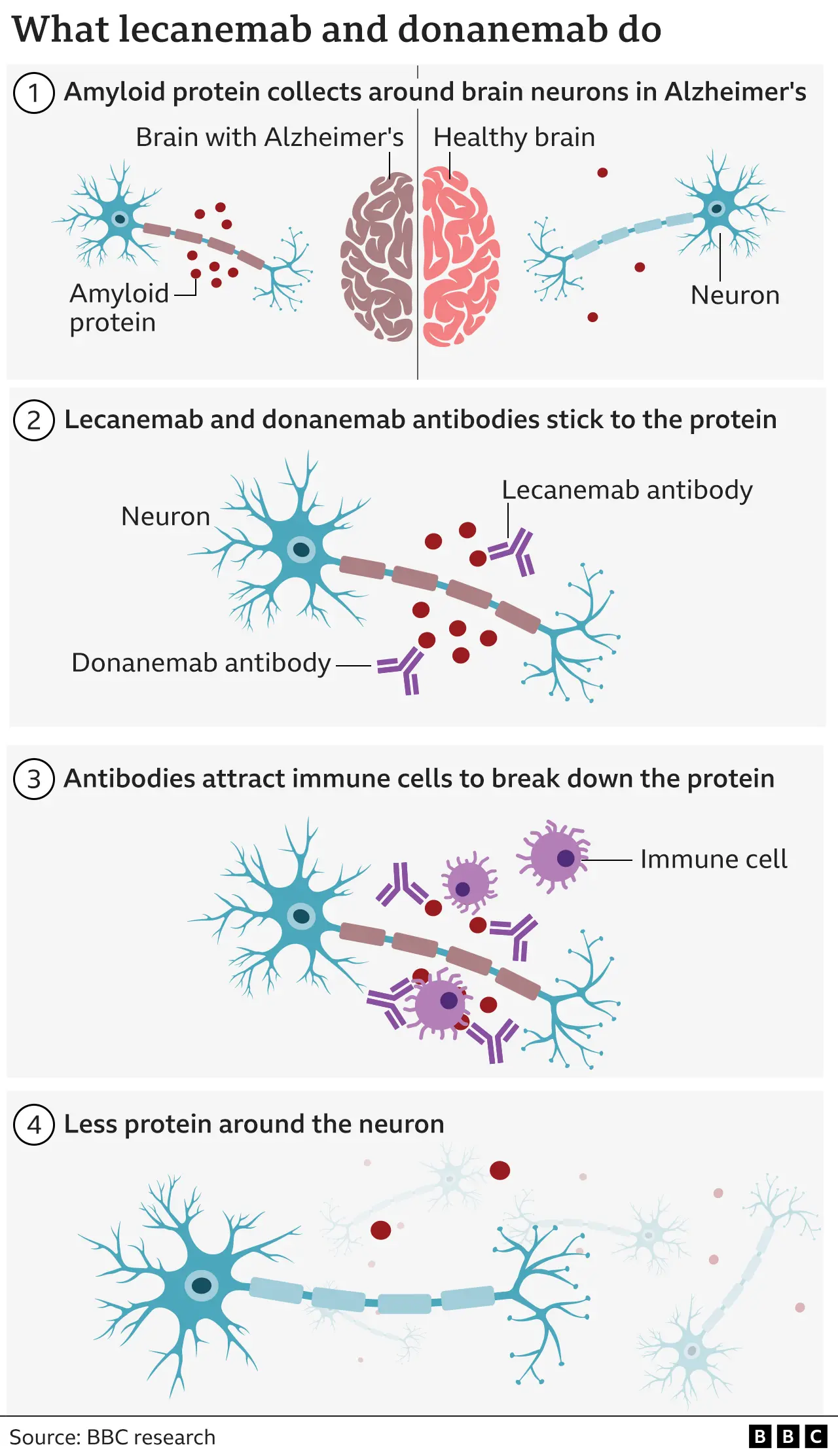
The goal is simple: clear the plaques, slow the cognitive decline, and preserve the patient’s quality of life. But the Cochrane analysis reveals a different story. After 18 months of treatment, the differences in memory and thinking abilities between those receiving the drugs and those receiving a placebo were minimal, if they existed at all.
Francesco Nonino, a lead author of the study from the IRCCS Institute for Neurological Sciences in Bologna, noted that while some earlier studies showed “statistically significant” results, these do not translate to “clinical relevance.” In plain English: a drug can move a number on a test score without actually making a patient’s life better.
The review found that in daily activities—the very things that define independence, such as managing finances or grocery shopping—the benefits were, at best, compact. For families dealing with the devastation of dementia, a “small” statistical advantage is cold comfort when compared to the reality of the disease’s progression.
Understanding the Risks: ARIA and Brain Swelling
The lack of significant benefit is only half of the equation. The other half is the risk profile. The Cochrane review highlights a concerning increase in side effects, specifically amyloid-related imaging abnormalities, known as ARIA.
ARIA can manifest as swelling of the brain or small hemorrhages. While the medical community monitors these closely, the presence of such side effects creates a precarious risk-reward ratio. When a medication provides minimal clinical improvement but carries the risk of brain swelling, the medical necessity of the treatment comes into question.
It is important to clarify that these antibody treatments are not intended for everyone. They are specifically targeted at people in the early stages of the disease, such as those with mild cognitive impairment (MCI) or early-stage dementia. This represents the window where researchers hoped the drugs could “brake” the progression of the illness.
The Landscape of Treatment in Germany
Despite the findings of the Cochrane review, some of these medications are already reaching patients. In Germany, Leqembi (Lecanemab) and Kisunla (Donanemab) have been available since Autumn 2025.
The process for accessing these drugs is rigorous. As of the risks and the specific target group, patients cannot simply receive a prescription. The protocol requires:
- A genetic test.
- Confirmed evidence of amyloid deposits via a PET scan or a cerebrospinal fluid (liquor) examination.
- Treatment administered exclusively in specialized medical centers.
For those in more advanced stages of the disease, the approach is different. Memantine is typically prescribed for moderate to severe Alzheimer’s dementia. Unlike the newer antibodies that try to remove plaques, Memantine works by protecting nerve cells from overstimulation by glutamate, a key neurotransmitter.
A Critical Look at Industry Funding
One detail from the Cochrane analysis that demands attention is the source of the data. Every single one of the 17 clinical studies evaluated was funded by pharmaceutical companies. While industry funding is common in drug development, it underscores the need for independent reviews like the one conducted by Cochrane to provide an unbiased interpretation of the results.
The tension here is between the pharmaceutical drive for innovation and the clinical reality for the patient. When the primary metric for success is the reduction of a protein (beta-amyloid) rather than the actual improvement of a human being’s ability to function, the results can be misleading.
Key Takeaways from the Review
- Study Scope: 17 clinical trials involving over 20,000 patients with early-stage Alzheimer’s.
- Clinical Impact: Minimal to no significant improvement in memory, cognition, or daily living activities after 18 months.
- Primary Risk: Increased incidence of ARIA (brain swelling and imaging abnormalities).
- Target Drugs: Lecanemab (Leqembi), Donanemab (Kisunla), Aducanumab, and Gantenerumab.
- Conclusion: The risks may outweigh the limited clinical benefits.
The fight against Alzheimer’s remains one of the most challenging frontiers in medicine. The disappointment surrounding these antibody treatments does not mean research has failed, but it does suggest that the “amyloid hypothesis”—the idea that simply removing plaques will stop the disease—may be incomplete or incorrect.
For now, the medical community must balance the desperation for a cure with the necessity of “do no harm.” As we have seen in sports and medicine alike, the most dangerous path is the one paved with hope that isn’t backed by hard, independent evidence.
The next critical checkpoint will be the emergence of new, independent longitudinal studies that are not funded by the manufacturers of these drugs. Until then, cautious optimism is the only responsible stance.
Do you believe the risk of brain swelling is acceptable for a slight chance of slowing cognitive decline? Let us understand your thoughts in the comments below.